Since 2014, ultra-high-dose rate irradiations or FLASH radiation therapy (FLASH-RT) has emerged as a promising optimization for radiotherapy treatments. Using a dose rate above 40 Gy s−1 provides tumor control equivalent to low-dose-rate irradiation while sparing healthy tissues (Favaudon et al 2014).
However, the mechanisms behind the FLASH effect are still not well understood, and the optimal dosimetric parameters (dose rate, dose, linear energy transfer (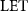 )) have not yet been defined (Hughes and Parsons 2020, Friedl et al 2022). Therefore, it is necessary to conduct studies to optimize this emerging dose delivery method.
)) have not yet been defined (Hughes and Parsons 2020, Friedl et al 2022). Therefore, it is necessary to conduct studies to optimize this emerging dose delivery method.
The study of the chemical and biological consequences of the FLASH effect, as well as the determination of the optimal dosimetric parameters, can only be carried out on irradiation platforms capable of ultra-high dose rate irradiations. Most of them use electron accelerators for their ability to deliver high beam intensities and their accessibility compared to other FLASH-ready devices such as synchrotron light sources and proton accelerators (Lin et al 2021, Schulte et al 2023).
However, proton beam therapy (PBT) offers several advantages for the delivery of FLASH irradiations. The clinical cyclotrons can deliver sufficiently intense beams and irradiate deep-seated tumors (Van De Water et al 2019, Jolly et al 2020). Furthermore, the high dose conformation offered by PBT could increase the healthy tissue sparing coming from FLASH-RT (Singers Sørensen et al 2022).
To overcome the limited availability of clinical cyclotrons for research purposes, several preclinical proton irradiation platforms have been set up (Diffenderfer et al 2022). These platforms, using beam energies that are much lower than those employed in the clinic, are dedicated exclusively to research and provide optimal irradiation conditions for conducting preclinical studies (Tillner et al 2014).
Since 2020, the Institut Pluridisciplinaire Hubert Curien (CNRS, University of Strasbourg) has developed the PRECy proton irradiation beamline, using 25 MeV protons accelerated by the CYRCé cyclotron, dedicated to in vitro and in vivo radiobiology studies (Bouquerel et al 2022).
This work presents, on the one hand, the validation of the Monte-Carlo simulation of the beamline through comparison with experimental measurements of energy and fluence, and on the other hand, the experimental validation of dose deposition. The simulation tool developed in this work can now be used to define the irradiation protocols required to meet experimental requirements. Additionally, post-irradiation dose monitoring using radiochromic films demonstrates the feasibility of delivering a precise dose for both conventional and FLASH dose rates.
2.1. PRECy platform2.1.1. CYRCé cyclotronCYRCé is an isochronous cyclotron TR24 (ACSI, Richmond, BC, Canada), designed at first for isotope production used in medical imaging. This cyclotron accelerates 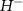 ions, which are converted to
ions, which are converted to 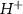 using a variable position stripper. The use of an external ion source allows access to the very low beam intensities needed in radiobiology experiments.
using a variable position stripper. The use of an external ion source allows access to the very low beam intensities needed in radiobiology experiments.
The extracted beam is ‘quasi-continuous’ with a bunch frequency of 85.085 MHz (one pulse of approximately 3.5 ns every 11.75 ns). The extracted beam intensity ranges from 100 fA up to 350 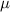 A and the beam energy can be selected between 16 and 25 MeV by changing the stripper position.
A and the beam energy can be selected between 16 and 25 MeV by changing the stripper position.
The cyclotron has two outputs, the first one is dedicated to the production of radioisotopes for nuclear medicine research (Johnson et al 2007) and the second is used to transport the beam to the irradiation beamline.
2.1.2. PRECy irradiation beamlineOnce extracted from the cyclotron, the beam is transported out of the bunker by the Cyclotron Beam Transfer Line (CBTL) to the research area, where beamlines are located. A switching magnet can deflect the beam in one of the two lines available (Bouquerel et al 2022). The PRECy irradiation line is one of these, dedicated to radiobiology experiments.
The purpose of this beamline is to deliver a homogeneous dose over a transverse irradiation field up to a 36.5 mm in diameter, for cell cultures and small animals’ treatment. The 25 MeV protons beam allow us to position the Bragg peak 6 mm deep within the tissues. A schematic diagram of this passive beamline is shown in figure 1.
Figure 1. Schematic diagram of the PRECy beamline. Proton beam shape is drawn in red.
Download figure:
Standard image High-resolution imageAt the entrance of the beamline, the transverse section of the beam is truncated by a centered circular collimator with an adjustable opening diameter ranging from 5 mm to 15 mm. The beam then passes through a scatterer composed of an aluminum sheet with a thickness adjusted to the desired irradiation field.
The beam next travels over 2.80 m to become sufficiently wide before passing through a second collimator, with an adjustable opening diameter that can be selected between 15 mm and 25 mm. This collimator retains only the central part of the scattered beam to obtain a uniform beam transverse profile over the desired irradiation field. However, most of the beam entering the beamline is lost due to scattering. For the scatterer used in FLASH irradiations (200 µm of aluminum), 90% of the incoming beam is lost and this fraction increases with the scatterer thickness.
Two homemade profilers are positioned to monitor the beam alignment. A Faraday cup, coupled with a Keithley 6517B picoammeter (Keithley Instruments LLC, Solon, OH, USA), can intercept the beam after the second collimator to measure its intensity. Irradiation time management is handled by the kicker or the shutter, which are described in section 2.1.5.
Once the beam is partially shaped, it is extracted in the air by going through a 50 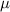 m thick aluminum foil, referred as the air exit to finally enter the conformation system as shown in figure 2.
m thick aluminum foil, referred as the air exit to finally enter the conformation system as shown in figure 2.
Figure 2. (a) Picture of the conformation system . (b) ‘Complete’ conformation system. (c) ‘Compact’ conformation system. In this figure, the beam is coming from the left.
Download figure:
Standard image High-resolution imageThis system, illustrated in figure 2(b), is composed of an energy degrader and a collimator. The energy degrader is a wheel containing 20 different thicknesses of aluminum from 0 mm to 2.761 mm allowing beam energy variation and spread out Bragg peak (SOBP) delivery (Constanzo et al 2019).
Following the degrader is the last collimator, which shapes the irradiation field according to experimental needs. For larger irradiations fields (i.e. diameter 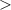 24 mm), the energy degrader is removed, and the collimator is coupled directly to the air exit, as shown in figure 2(c).
24 mm), the energy degrader is removed, and the collimator is coupled directly to the air exit, as shown in figure 2(c).
When beam energy is reduced using the degrader, significant beam scattering is introduced. In this case, it is important to select particles with the smallest scattering angles to ensure a uniform transverse dose profile. To achieve this, the degrader is positioned 3 m upstream of the irradiation field, at the scatterer, to take advantage of the beamline’s collimation system, which allows for the selection of the desired angles. For in vitro studies, 48- to 6-well culture plates (with well diameter ranging from 11.05 mm to 34.80 mm) are mounted vertically on a remote-controlled sample holder, allowing sequential irradiation of each well.
2.1.3. Dose calculationThe deposited dose D (in Gy) for a passive irradiation is calculated with the following equation:

where φ is the beam fluence, 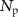 the total number of protons received by the sample during irradiation,
the total number of protons received by the sample during irradiation,  the surface of the irradiation field,
the surface of the irradiation field, 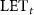 the fluence-averaged LET and ρ the density of the irradiated medium. In the rest of this study, the dose is calculated in water.
the fluence-averaged LET and ρ the density of the irradiated medium. In the rest of this study, the dose is calculated in water.
If we consider a constant proton beam intensity 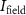 at the irradiation field over the irradiation time
at the irradiation field over the irradiation time 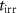 ,then equation (1) becomes:
,then equation (1) becomes:

where e is the value of the elementary electric charge.
2.1.4. Irradiation protocolPrior to each irradiation session, the cyclotron parameters and beam alignment in the beamline are optimized to ensure beam stability and a uniform fluence rate in the transverse irradiation plane. For each beamline configuration used, a calibration factor to convert the beam intensity measured by the Faraday cup in the beamline 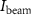 to the beam intensity in the irradiation plane
to the beam intensity in the irradiation plane 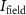 is measured using a second removable Faraday cup placed in the position occupied by the sample. During irradiation, no online dose control is performed, requiring a precise characterization of the parameters involved in equation (2) to ensure an accurate dose delivery.
is measured using a second removable Faraday cup placed in the position occupied by the sample. During irradiation, no online dose control is performed, requiring a precise characterization of the parameters involved in equation (2) to ensure an accurate dose delivery.
Beam delivery and the automatic sample holder are managed by an homemade control software. Before each irradiation, the intensity of the beam required to achieve the desired dose rate 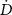 is determined using equation (3), derived from equation (2) :
is determined using equation (3), derived from equation (2) :

The cyclotron source is then adjusted to generate a beam with sufficient intensity to reach 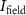 . For an in vitro irradiation using a 24 mm-diameter collimator, with an
. For an in vitro irradiation using a 24 mm-diameter collimator, with an 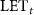 in the sample (considered as water) equals to (2.74 ± 0.03) keV (beam energy = 18.73 MeV, energy dispersion = 180 keV), a conventional dose rate of 2 Gy min−1 is reached with an
in the sample (considered as water) equals to (2.74 ± 0.03) keV (beam energy = 18.73 MeV, energy dispersion = 180 keV), a conventional dose rate of 2 Gy min−1 is reached with an 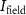 value of 5.50 pA. For a FLASH dose rate of 100 Gy s−1,
value of 5.50 pA. For a FLASH dose rate of 100 Gy s−1, 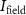 must be increased to 16.51 nA. During radiobiology experiments, the beam intensity extracted from the cyclotron remain much lower than the ones used for radioisotope production.
must be increased to 16.51 nA. During radiobiology experiments, the beam intensity extracted from the cyclotron remain much lower than the ones used for radioisotope production.
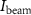 is measured by the Faraday cup in the beamline to determine
is measured by the Faraday cup in the beamline to determine 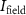 . Then, the irradiation time
. Then, the irradiation time 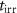 is calculated by the control software, according to the required dose. The irradiation time is ensured using the kicker and/or the shutter described in the following paragraph. After the irradiation, the Faraday cup is lowered again to verify that no drift in beam intensity has occurred. If this is not the case, the delivered dose is recalculated using an average beam intensity between the measurements taken at the beginning and the end of the irradiation. For short irradiation times as the ones used in FLASH, we did not observe any drift in beam intensity.
is calculated by the control software, according to the required dose. The irradiation time is ensured using the kicker and/or the shutter described in the following paragraph. After the irradiation, the Faraday cup is lowered again to verify that no drift in beam intensity has occurred. If this is not the case, the delivered dose is recalculated using an average beam intensity between the measurements taken at the beginning and the end of the irradiation. For short irradiation times as the ones used in FLASH, we did not observe any drift in beam intensity.
The uncertainty on dose calculation associated to this protocol is given using the following equation:
 2.1.5. Irradiation time management
2.1.5. Irradiation time managementA radio-frequency or RF kicker (figure 3(a)), located between the source and the accelerator cavity, controls the irradiation time and ensures rapid beam shut-off (Pellicioli et al 2023). It consists of two parallel tungsten plates to which an alternating voltage up to 8 kV is applied, located between two collimators. The voltage between the plates is supplied by an RF system operating at a frequency equal to one-fourth of the cyclotron frequency. As a result, the beam is transmitted by bunches at a frequency of 42.5425 MHz, which is half the cyclotron frequency.
Figure 3. (a) Picture of the RF kicker. (b) Mechanic scheme of the shutter.
Download figure:
Standard image High-resolution imageMoreover, by applying an appropriate phase to the RF system, it is possible to cut off the beam. Beam rejection of up to 99.998% can be achieved using this device.
To increase beam rejection, a mechanical shutter (figure 3(b)) is present in the CBTL. It consists of an extruded aluminum block connected to a motorized translation axis. By rotating the extruded block, a complete rejection is achieved. However, the trade-off is an increase of beam rise and fall times compared to the RF kicker.
2.1.6. Fluence management in FLASH conditionsFor Ultra-high dose rate irradiations, the finite rejection of the kicker leads to extra-dose deposition (up to 0.2 Gy at 100 Gy s−1) due to the time needed to set up and remove the irradiated sample. In this case, both kicker and shutter are used, as shown in figure 4.
Figure 4. Irradiation scheme in FLASH conditions.
Download figure:
Standard image High-resolution imageThe kicker handle irradiation time and the shutter open for a slightly longer time. With this configuration, we have access to both precise irradiation time and minimize dose deposition between irradiations.
2.2. GATE simulationIn order to deliver the required dose using equation (2), the 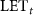 has to be correctly evaluated. The equation of the
has to be correctly evaluated. The equation of the 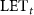 is given by (Kalholm et al 2021):
is given by (Kalholm et al 2021):

where 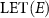 and
and 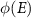 are the LET and the fluence of protons with a kinetic energy E. The energy spectrum of the beam and the configuration of the beamline can be the input of a Monte-Carlo modeling to evaluate the
are the LET and the fluence of protons with a kinetic energy E. The energy spectrum of the beam and the configuration of the beamline can be the input of a Monte-Carlo modeling to evaluate the  . We implemented such a Monte Carlo simulation on the GATE platform version 9.1 (Sarrut et al 2014).
. We implemented such a Monte Carlo simulation on the GATE platform version 9.1 (Sarrut et al 2014).
This simulation includes the beam transportation in the PRECy beamline (figure 1), modeled with its optical properties (Bouquerel et al 2019) using the Pencil Beam class, the conformation system (figure 2) and the sample to be irradiated. The simulation has been parametrized to have an accurate proton transport inside the setup (Grevillot et al 2010). Simulation parameters and beam optical properties are presented respectively in tables 1 and 2 below.
Table 1. Parameters used for the GATE simulation. 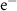 denotes an electron,
denotes an electron, 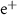 a position, γ a photon and
a position, γ a photon and 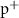 a proton.
a proton.
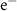 ,
,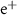 ,γ)0.025 mmMaximum step (
,γ)0.025 mmMaximum step (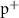 )0.020 mm
)0.020 mmTable 2. Optical parameters used for the Pencil Beam class. The beam is considered symmetric with respect to the x and y axes. σx and σθ represent the standard deviation of the normal probability density function for the position and the divergence (determined using unscattered beam profiles), respectively, while ε denotes the beam emittance (measurements from (Bouquerel et al 2019)).
ParameterValueσx1.67 mmσθ0.890 mradε4.40 mm.mrad2.3. Instrumentation for beam characterization2.3.1. Measurement of proton energy spectrumFor this measurement, a four stacked depleted silicon detectors telescope was used (AMETEK Inc. Berwyn, PA, USA), each with a different sensitive thickness placed either in air or inside a vacuum chamber. The telescope configuration is presented in figure 5(b).
Figure 5. (a) Picture of the detectors inside the vacuum chamber. (b) Schematic of the telescope.
Download figure:
Standard image High-resolution imageEach detected event is processed by a FASTER acquisition system developed by the LPCC lab (https://faster.in2p3.fr) and recorded with its timestamp and the amplitude of the signal measured, proportional to the energy deposited by the incident proton.
Once the detectors have been calibrated using a self-calibration protocol (Constanzo et al 2019), the coincidences can be reconstructed and the energy of the incident protons measured. For measurements inside the vacuum chamber (figure 5(a)), the incident proton energy is the sum of the energy deposited in the four detectors. For measurements in air, an additional calculated term must be added, corresponding to the energy lost in the air between the detectors.
2.3.2. Measurement of the fluence deliveredAs stated in equations (1) and (2), the fluence φ is given by :

To measure the number of protons delivered, we used a BC-420 plastic scintillator (Luxium Solutions, Hiram, OH, USA) with a decay time of 1.4 ns, coupled to a Hamamatsu H7415 photomultiplier tube (Hamamatsu Photonics K K, Hamamatsu, Shizuoka, Japan). Scintillator dimensions are large enough to cover the entire irradiation field. A FASTER-QDC card is used to measure the charge above the specified threshold and record it with its timestamp.
Figure 6 shows a histogram of the events detected by the plastic scintillator as a function of the measured signal, for an irradiation at a dose rate of 2 Gy min−1, with a beam energy of 24.16 MeV and an energy dispersion of 100 keV. With an energy resolution of 13% at 24.16 MeV, it is possible to distinguish the signal generated by a single proton from that produced by a pile-up of multiple protons. Using manually set thresholds to separate the different signal stacks, it is possible to count the number of incident protons during irradiation.
Figure 6. Histogram of the measured signal. Black numbers in the figure correspond to the number of protons in the events between the two thresholds.
Download figure:
Standard image High-resolution imageThe reproducibility of the number of protons 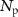 delivered during an irradiation is assessed by counting the amount of protons delivered for multiple irradiations following the protocol described in section 2.1.4.
delivered during an irradiation is assessed by counting the amount of protons delivered for multiple irradiations following the protocol described in section 2.1.4.
The relative dispersion of these measurements, 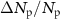 , may arise from fluctuations in beam intensity and irradiation time over the irradiation period:
, may arise from fluctuations in beam intensity and irradiation time over the irradiation period:
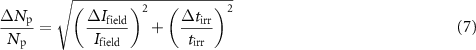 2.3.3. Measurement of the irradiation time
2.3.3. Measurement of the irradiation timeThe BC-420 scintillator was also used to measure beam rise and fall times for both kicker and shutter to confirm our control of irradiation time, including under FLASH conditions. A histogram representing the counting rate over time is then fit by the following function:
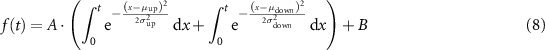
Where 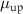 and
and 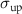 correspond respectively to the centroid and the standard deviation of the erf function fitted to the rise of the beam, while
correspond respectively to the centroid and the standard deviation of the erf function fitted to the rise of the beam, while 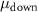 and
and 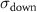 are the parameters of the erf function fitted to the beam fall. A and B are two parameters for selecting the amplitude and the baseline of the function.
are the parameters of the erf function fitted to the beam fall. A and B are two parameters for selecting the amplitude and the baseline of the function.
Using the fitted function, we measure the rise time 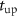 as the time elapsed between 10% and 90% of maximum, and the fall time
as the time elapsed between 10% and 90% of maximum, and the fall time 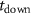 as the time elapsed between 90% and 10% of the maximum.
as the time elapsed between 90% and 10% of the maximum.
The total irradiation time 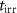 is measured as the difference between
is measured as the difference between 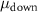 and
and 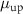 . The difference
. The difference 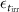 between
between 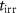 and the time defined by the control software is used to calculate the error on irradiation time :
and the time defined by the control software is used to calculate the error on irradiation time :

When using the shutter, the rise and fall times are long enough to be measured using a single irradiation, which is not the case for the RF kicker. For the latter, several irradiations of identical duration are added together, using the signal sent to the kicker by the control software as a time reference.
2.3.4. Control of the delivered doseThe dose delivered in the transverse irradiation field is controlled using radiochromic films. Two types of film were used in this study: Gafchromic EBT3 (Ashland, Covington, GA, USA) and Orthochromic OC-1 films (Orthochrome, Bridgewater, NJ, USA). While EBT3 films are the reference films used for dose monitoring, their response is dose rate-dependent, which is not the case for OC-1 films (Villoing et al 2022). The latter will therefore be used for dose monitoring under FLASH conditions.
The film response to irradiation is measured using the net optical difference netOD (Reinhardt et al 2015):

where 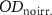 and
and 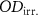 are the optical densities for unirradiated and irradiated films respectively.
are the optical densities for unirradiated and irradiated films respectively.
The films wer scanned using an EPSON Perfection V700 scanner (EPSON, Suwa, Nagano, Japan) with a sampling of 360 dots per inch (i.e. 70.56 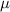 m of spatial resolution) and each color encoded over 16 bits. For EBT3 films, the film is scanned before and 24 hours after irradiation in order to calculate netOD. For OC-1 films,
m of spatial resolution) and each color encoded over 16 bits. For EBT3 films, the film is scanned before and 24 hours after irradiation in order to calculate netOD. For OC-1 films, 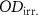 is measured 24 hours after irradiation, and
is measured 24 hours after irradiation, and 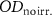 , measured in a non-irradiated region around the irradiation field, is also recorded 24 hours after irradiation. A preliminary experiment has shown that this protocol improves response stability compared with the method used for EBT3 films.
, measured in a non-irradiated region around the irradiation field, is also recorded 24 hours after irradiation. A preliminary experiment has shown that this protocol improves response stability compared with the method used for EBT3 films.
For each experiment, a calibration curve is performed with the following function where a, b and c are determined on a set of known dose measurements:

Regarding the 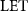 quenching correction, the literature reports a relative efficiency (RE) value of 0.96–0.97 for the
quenching correction, the literature reports a relative efficiency (RE) value of 0.96–0.97 for the 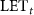 values in this study (Sanchez-Parcerisa et al 2021). We opted not to apply this correction for several reasons. First, this RE value is close to 1 (indicating no significant quenching effect). Second, our calibration curves were made at
values in this study (Sanchez-Parcerisa et al 2021). We opted not to apply this correction for several reasons. First, this RE value is close to 1 (indicating no significant quenching effect). Second, our calibration curves were made at 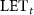 values identical or very close to those used in this study. Finally, the uncertainties associated with the RE measurements are not negligible compared to the RE value.
values identical or very close to those used in this study. Finally, the uncertainties associated with the RE measurements are not negligible compared to the RE value.
To guarantee the irradiation conditions when using the PRECy beamline, we have characterized all the parameters that have an impact on dose deposition such as the beam energy, the stability of beam intensity and the irradiation time. These results were used to validate a GATE simulation of the irradiation line. Finally, we have assessed the uniformity of the dose delivered with respect to irradiation field size and
Comments (0)